PCL compost (lab)
PCL soil (lab)
PCL soil (field)
Biodegradable polymers are a complex topic, and how long a specimen takes to fully degrade depends on many factors. For the aliphatic polyesters, temperature is a primary variable, with biodegradation proceeding roughly 2-3 times more quickly for every 10C increase in temperature. This is why hot-composting at around 60C is the recommended disposal method - at these temperatures, biodegradation proceeds up to 50 times faster than in colder conditions. (The PGA manufacturer Kureha nicely illustrates this temperature dependence with the hydrolysis of a Kuredux sphere.)
For biodegradation to occur at all, aliphatic polyesters generally require the assistance of certain microbes, together with temperatures around or exceeding the material's glass transition point. Accordingly, microbes capable of catalysing the biodegradation of PCL (Tg of -60C) can be found in the open environment, whereas those required to break down PLA tend to only thrive in much hotter conditions (due to PLA's higher Tg of +60C).
The commonly cited compostable test standards EN13432/14995 and ASTM D-6400 take this into account by specifying composting conditions around 58C. This allows a wider range of products to be classed as a biodegradable/compostable. However, meeting the requirements of these standards does not indicate that the product will also readily biodegrade at significantly lower temperatures.
Important note: The above test standards are strictly for products, not materials. Many products are sold on the basis of being made from materials that claim to be "biodegradable" or "compostable". Supporting evidence for the actual product being biodegradable is often lacking.
Published Research Results
Although there are other biodegradable test standards available, all are necessarily artificial since they need to be reproducible. It is an acknowledged problem that laboratory testing can never properly represent the diverse natural environments into which biodegradable plastic products may be disposed.
Ideally, anyone considering using "biodegradable" plastic products should test them in the actual intended environment. For short-term applications, any product that is rapidly biodegradable should show some evidence of degradation within a few months of exposure.
Of course, this is not always practical, and over the past 30 years there have been many attempts to "fill in the gaps" and evaluate biodegradable polymers under various real-world conditions. Due to time constraints, much of this testing has been done on pieces of thin film (typically <100μm), and this is understandable as many of the published research papers are actually conducted by university students pursuing Masters or PHD qualifications. Thin films are relatively easy to produce in a university laboratory by solution casting, and they degrade quickly enough to be evaluated within the timeframe of a typical degree. (They can also be easily compared to a cellulose control specimen - i.e., a piece of paper.)
As well as a visual inspection of any degradation (often done with SEM), weight loss, tensile strength testing (stress & strain) and infra-red spectroscopy (FTIR) are all commonly used techniques to characterise the test pieces. FTIR conveniently identifies changes to the underlying molecular structure, and this is frequently supplemented by calorimetry measurements (DSC) that reveal changes in bulk crystallinity. Access to a biochemical laboratory permits analysis and identification of primary degraders, and many microbial strains capable of degrading polymers under different conditions are identified in the literature.
Academic access to injection moulding facilities is, or course, somewhat rarer, so there are relatively few published papers on the behaviour of bulkier samples. (Such samples would be more representative of actual products that might be advantageously made from biodegradable plastics.) This gap is slowly closing and recent biodegradation research conducted at the University of Manchester (Al Hosni, Pittman & Robson, (2019)) was unusual in that it tested much thicker 3mm samples of PCL and PLA rather than just thin films.
These plaques were tested over a two year period in both controlled laboratory and uncontrolled outdoor environments, and their results confirmed much of the previous research done on these materials - namely that PCL biodegrades rapidly under all conditions, whereas PLA biodegrades much more slowly. A simplified representation of their weight-loss results is presented below. Please consult their original paper for full details.
[Rapstrap were not involved in this particular research, but we would be happy to collaborate with future studies.]
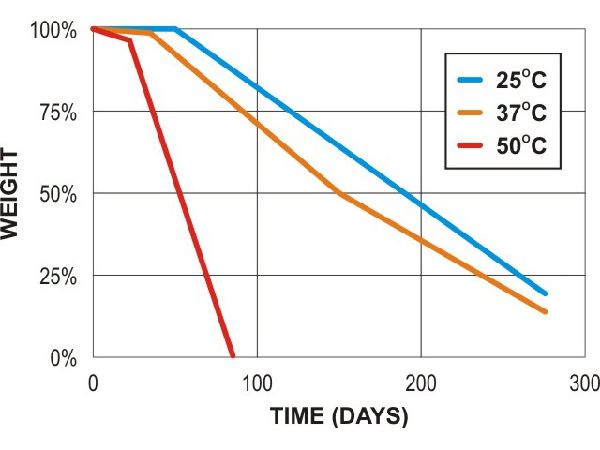
Poly caprolactone (PCL - 3mm test plaques)

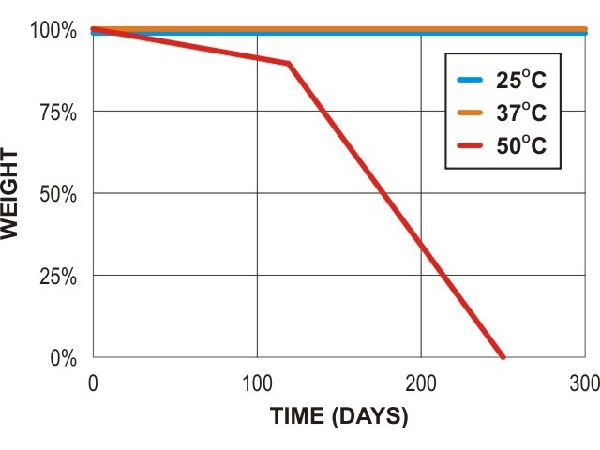
PCL compost (lab)

PCL soil (lab)

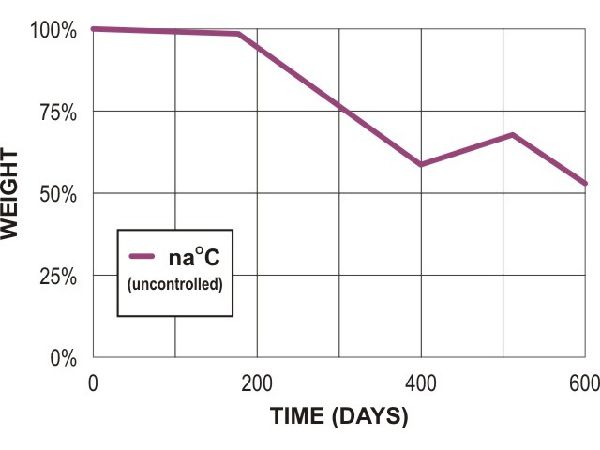
PCL soil (field)
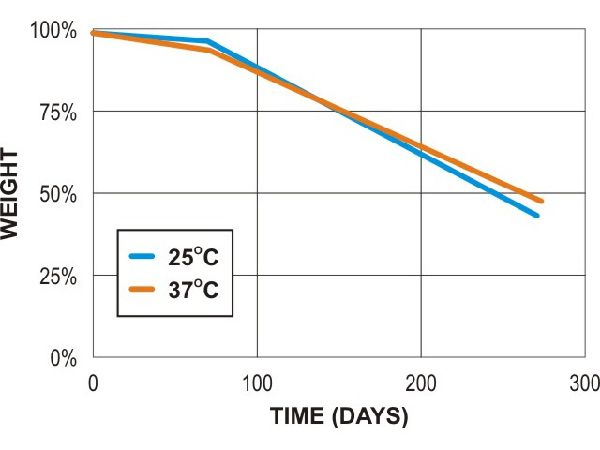
Poly lactic acid (PLA - 3mm test plaques)
PLA compost (lab)
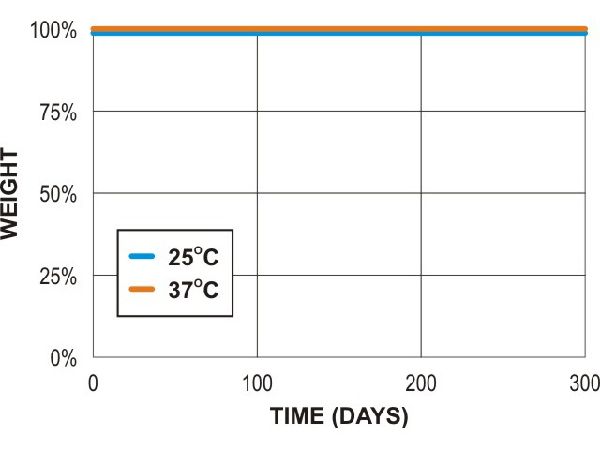
PLA soil (lab)
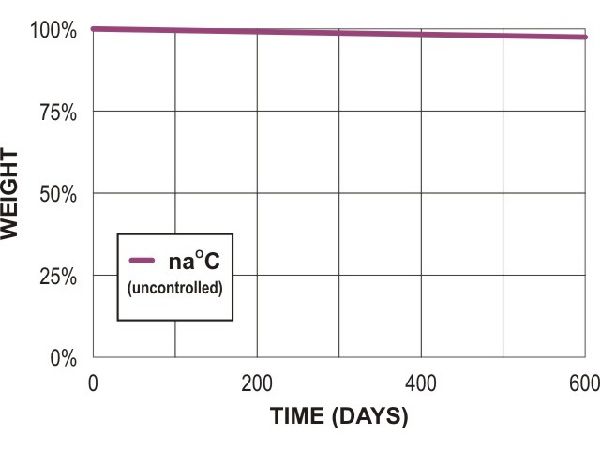
PLA soil (field)
Adapted from Al Hosni, A. S. K., Pittman, J., & Robson, G. (2019) figures 1-3. (University of Manchester)
“Microbial degradation of four biodegradable polymers in soil and compost, demonstrating polycaprolactone as an ideal compostable plastic.”
Waste Management, 97, 105-114.
https://doi.org/10.1016/j.wasman.2019.07.042